How fast does a shield get uncomfortable when you use it to block a gout of dragon’s flame?
This is the third of five posts answering that burning question (pun intended). So far we’ve learned that:
In this post we’ll talk about Watts, shields, and microwaves. Let’s get to it!
This is the third of five posts answering that burning question (pun intended). So far we’ve learned that:
- Dragon flame heats a shield using convection and radiation (and probably some other stuff that we’re ignoring for now) - see Part 1.
- Convection probably dumps about 1,100 Watts of heat into the shield (at least at first) - see Part 2.
In this post we’ll talk about Watts, shields, and microwaves. Let’s get to it!
Dragon's Fury, art by BenWootten, deviantart.com
First, what are Watts?
You’ve probably seen them listed on light bulbs, microwaves, and electric bills. Watts are a measure of energy per second (1 Watt = 1 Joule / second). Energy comes in a lot of forms – electrical, potential, kinetic – but electric energy shows up most in spec sheets. Some things are rated by usage (a 60-Watt bulb uses 60 Joules every second), while others are rated by output (my 1000-Watt microwave puts 1000 Joules into my food every second, though it uses 1,680 Joules of electrical energy every second to do it).
So we already have a pretty good idea of how fast convection is dumping heat (energy) into our knight’s shield… pretty close to the same rate that your microwave dumps heat into your hot pocket.
But how fast does that raise the temperature of the shield? How do we relate energy (Joules) to a change in temperature?
Glad you asked. Specific heat capacity is a property that tells you how many Joules it takes to heat a gram of something up by 1 degree C. It’s different for different materials, and it’s not always what you’d expect (it takes only 0.1 Joules to heat up a gram of lead by 1 C, while it takes over 4 Joules to heat up a gram of water by the same amount). Here’s a nice table of specific heat capacities.
Our knight’s shield is probably steel (actually metal shields were pretty rare – leather or wood was much more common – but lets assume our knight wasn’t dumb enough to bring a wooden shield to a dragon fight). It’s probably 2mm thick (typical for plate armor). Last time we assumed it was 1 meter (40 inches) square (pretty huge for a shield, but maybe enough to protect a whole knight). That gives us enough info to calculate the mass of the shield:
You’ve probably seen them listed on light bulbs, microwaves, and electric bills. Watts are a measure of energy per second (1 Watt = 1 Joule / second). Energy comes in a lot of forms – electrical, potential, kinetic – but electric energy shows up most in spec sheets. Some things are rated by usage (a 60-Watt bulb uses 60 Joules every second), while others are rated by output (my 1000-Watt microwave puts 1000 Joules into my food every second, though it uses 1,680 Joules of electrical energy every second to do it).
So we already have a pretty good idea of how fast convection is dumping heat (energy) into our knight’s shield… pretty close to the same rate that your microwave dumps heat into your hot pocket.
But how fast does that raise the temperature of the shield? How do we relate energy (Joules) to a change in temperature?
Glad you asked. Specific heat capacity is a property that tells you how many Joules it takes to heat a gram of something up by 1 degree C. It’s different for different materials, and it’s not always what you’d expect (it takes only 0.1 Joules to heat up a gram of lead by 1 C, while it takes over 4 Joules to heat up a gram of water by the same amount). Here’s a nice table of specific heat capacities.
Our knight’s shield is probably steel (actually metal shields were pretty rare – leather or wood was much more common – but lets assume our knight wasn’t dumb enough to bring a wooden shield to a dragon fight). It’s probably 2mm thick (typical for plate armor). Last time we assumed it was 1 meter (40 inches) square (pretty huge for a shield, but maybe enough to protect a whole knight). That gives us enough info to calculate the mass of the shield:
From our table of specific heats, we know that steel requires 466 Joules to heat 1 kg by 1 degree C, so if we’re dumping 1,100 Joules per second into it:
So the shield heats up by 0.15 C every second. At that rate, it would take almost 6 minutes of dragon fire to heat the knight’s shield up to an uncomfortable 75 C (167 F).
Not very impressed? Me either.
But wait! Next time we look at radiation, which is much more impressive (and also much more likely to cook our brave knight).
Question? Comment? Something in Science Fiction or Fantasy you'd like to see analyzed? Let me know!
Not very impressed? Me either.
But wait! Next time we look at radiation, which is much more impressive (and also much more likely to cook our brave knight).
Question? Comment? Something in Science Fiction or Fantasy you'd like to see analyzed? Let me know!

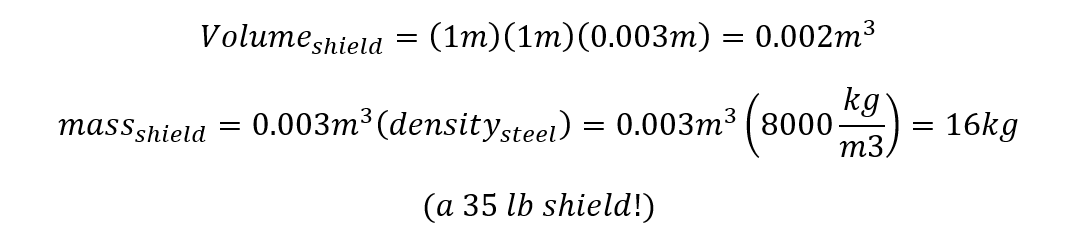
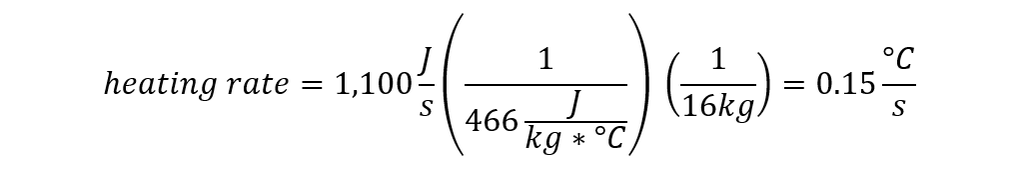
 RSS Feed
RSS Feed

